Low- and middle-income countries (LIC/MICs) face severe challenges due to limited economic opportunities. In addition to the economic struggles, LICs also bear a large burden of transmittable diseases, posing severe risks to the population's wellbeing.[1] Healthcare systems and healthcare providers in LICs are often ill-equipped to treat the patients in the best possible way, especially in rural areas. Given the economical and infrastructural challenges in LIC, PoC diagnostics, which are often characterized by being independent of laboratory or medical infrastructure, as well as being highly affordable and holding considerable promise to improve the situation. Yet the actual commercialization of PoC diagnostic-tests lags well behind the innovative research and developments done in laboratories.
Several review articles on the topic have been written in past years, but most are specialized on one specific aspect, logistical shortcomings,[2] funding and collaboration considerations,[3] technological standpoints,[4] or usage profiles from home to laboratory.[5] This review investigates the topic from a different angle: it looks at the barriers of PoC diagnostics along the entire value chain, from the first idea in a laboratory to the use of the final product by a healthcare provider. The aim is to locate the "leaks in the pipeline" of PoC commercialization, where PoC devices do not manage to proceed to a next step in the value chain. The value chain is separated into three distinct domains, research, market, and usage, each consisting of separate steps.
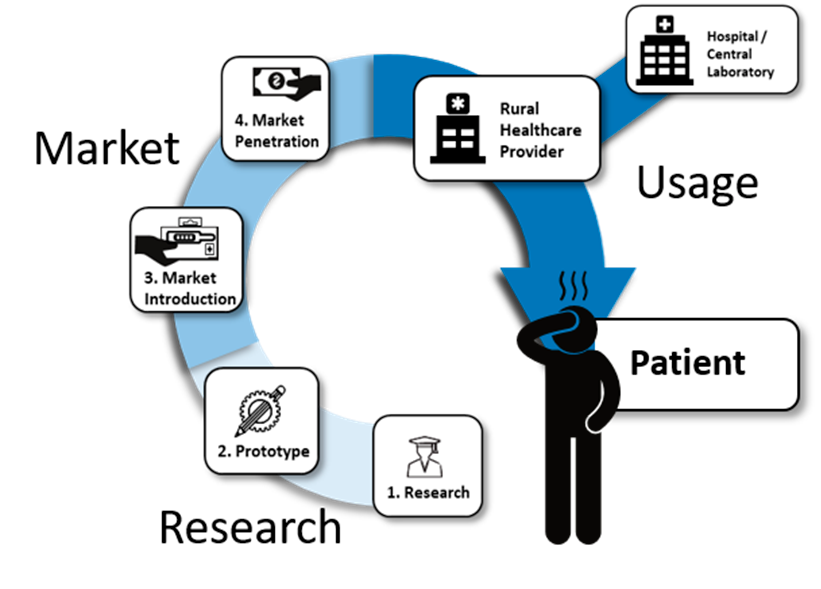
Research
Fundamental Research: Funding Availability and Focus
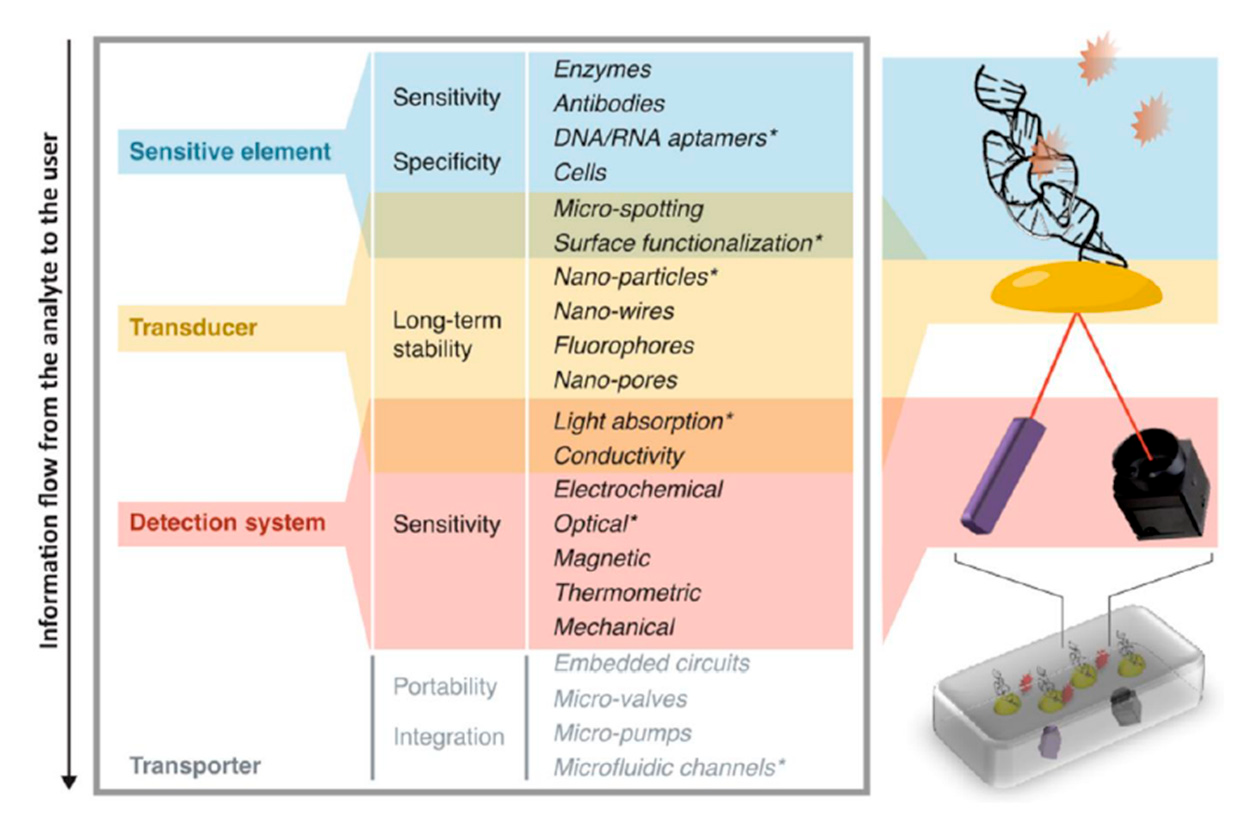
Fundamental research is the first step in the development of a PoC device. Historically, the research of PoC devices started with simple dipstick tests with immobilized reagents, for example, for the detection of glucose.[6] Later the laboratory success of immunoassays, especially the high sensitivity of radioimmunoassays and enzyme-linked immunoassays, created interest in improving those methods into rapid tests, which eventually became lateral flow immunoassay, the most abundant type of PoC device.[7] While lateral flow devices are the most common, the trend in research goes towards devices with higher complexity, able to handle more complicated samples, multiplex and detect challenging analytes.[6] The greater complexity is shown in their mechanisms of action, which are often optical, electro(chemical) or magnetic,[8] but also include other modes, e.g., thermal sensor systems.[9,10] Optical systems range from UV/Vis or fluorescence sensors to quantum dot and surface-plasmon-resonance technology or even genetically encoded biosensors.[11–14] Electro-(chemical) systems range from basic ampere or voltametric systems over graphene-based field-effect-transistors to DNA-annealing-based redox-reporter assays.[15–18] However, compared to the abundance of new research, the actual commercialization lags behind and market examples welcoming these new innovations are rare.[19]
Choosing a Suitable Design Philosophy
There are several factors that may misguide new developments, an important one being the design philosophy of new devices. Research in high-income countries (HIC) is rarely aimed at inexpensive technologies with wider impact,[4,20] which are direly needed in resource-limited settings. Instead, research in HICs tends to focus on high-efficiency devices with even higher complexity, which is not a problem for affluent countries with good infrastructure; however, due to their complexity, such devices are often not usable (or affordable) in LICs.[21] Several studies point out the differences in approach between high-complexity PoC diagnostic test platforms (HCTs), such as the GeneXpert and low-complexity PoC tests (LCTs), such as widely available lateral flow tests.[4,20,21] While HCTs can conduct more difficult and sensitive diagnostic tests, they are also more complex and thus require more training, maintenance and infrastructure. LCTs, on the other hand, lack sensitivity and diagnostic power, but are highly affordable and low maintenance.
A stark design-philosophy mismatch emerges: in HICs the most suitable biomarker for a given illness is chosen without much regard for infrastructure. Venipuncture, for example, is a given in HICs, which means tests developed have no strong restrictions for the needed sample size. This can pose a problem for LICs in rural settings without a trained phlebotomist, venipuncture is not possible and the test has to be able to work either with easier to acquire samples, such as sputum or urine, or the much smaller blood sample quantities of a finger prick or heel-stick samples. Heel-stick sample sizes are usually under 5% of the size of venipuncture samples.[22] Instead of building a system around the best biomarker, the system needs to be created around the available infrastructure first, which is a considerably different design philosophy.[22,23]
Taking Aim: Proper Target Analytes
The target of research is also different in HICs and LICs. Resource-rich settings do not focus on research on neglected tropical diseases (NTDs); their burden is mostly nonexistent in affluent countries, and they are therefore less interesting for commercial research, as the chance of return of investment is limited.[24] From 1975 to 2004, only 1% of drugs were developed for NTDs (21 out of 1556).[3] Other gaps in knowledge might be easier to miss, for example, local differences in diseases, such as geographical variability in antigen presentation and DNA/RNA signals. Immunosuppression due to HIV leads to lower host response signals and reduces the sensitivity of some nucleic acid tests for the detection of pulmonary TB.[25,26] Research can only make an impact in LICs if a bottom-up approach is used that takes the infrastructure and environment of LICs into account from the start.[4]
Funding in LICs
Research in LICs themselves is beneficial as it focuses directly on the regional circumstances and problems, with a LICs-centered philosophy. However, PoC funding in LICs themselves is highly inconsistent;[20,27] this leads to dependence on other funding opportunities, such as NGOs and development partnerships. This is not only the case for funding of PoC-research, but also for treatment-research and treatment itself. For some countries in Sub-Saharan Africa, HIV expenditures are strongly reliant on external sources, despite this being one of the region's most important health risks. Kenya and Uganda contribute less than 15% of funding to their own national HIV-relief efforts, Mozambique even contributes only 3%.[20,27] In cases where there is secure funding through outside sources, it naturally tends to focus on treatment or prevention, while diagnostic research receives much less funding.[4,28]
Incentives to Change Focus in HICs
New programs are being implemented to incentivize product development for neglected tropical diseases.[3] Examples include the "Priority Review Voucher" (PRV) program in the United States, which gives out transferable priority FDA-review vouchers, an implemented "pull-mechanism" to motivate development in neglected areas. Push-incentives include the Global Health Investment Fund (GHIT), a partnership connecting the Japanese Government, NGOs, and large drug and diagnostic manufacturers, targeting poverty-connected diseases and NTDs.[3,31] Other named initiatives are the "Global Health Investment Fund (GHIF)" and the "Wellcome Trust Pathfinder Award".[3] NGOs, e.g., the Bill & Melinda Gates Foundation, have contributed a substantial amount of funding for NTDs research.[32–36]
To change the philosophy of research, "Frugal Development" must be considered from the start to build devices with relatively low complexity that are not only affordable and usable in LICs settings but also maintainable and repairable.[37–40] 3D-printing and other new technologies may help create spare parts on demand; these new devices have already been shown to create simple microfluidic parts and supply hospitals with respirator valves during the COVID-19 pandemic.[41,42]
Proof-of-Concept and Prototypes: The Importance of Appropriate Device Characteristics
Device Characteristics
Engineers in affluent countries tend to design devices that assume HICs infrastructure standards, meaning well-funded laboratories in regulated environments with quality control. This can be problematic in LIC, and therefore special design considerations must be taken into account.[20] Devices that perform well in controlled settings often fail when challenged with tropical conditions in LICs.[26] Especially in rural settings, access to electricity might be problematic in terms of powered PoC devices,[22,26,43] but also a lack of cold storage options can form a significant challenge.[9,22,23,43] Limited refrigeration and power supply therefore demands that the device and its disposables have to be stable in the long term, even at high temperatures, while powered PoC devices need to be able to run on battery or solar.[4,26,44]
Being ASSURED
Many authors agree that beneficial device characteristics follow the WHO guidelines for PoC diagnostics, symbolized by the acronym ASSURED: Affordable, Sensitive, Specific, User-friendly, Rapid and robust, Equipment-free and Delivered to those who need them.[20,22,45–48] These requirements are regularly mentioned, but it has to be noted that these are ideal and strong requirements that only a selected few devices can meet.[21]
While progress has been made on PoC tests for syphilis, chlamydia, and gonococcal infections, there is still not one test that complies with all ASSURED criteria.[46] Sensitivity and selectivity get increasingly more difficult to achieve the closer the system gets to a "perfect" system with an accuracy of 100%.[21,22,26] In rural LICs settings, a PoC diagnostic tool needs to be small, portable and highly affordable.[43] While many authors put great emphasis on affordability, others argue that Zero-Cost is not an important parameter; reliability and standardization are seen as more crucial, and it might be misguided to assume that "poverty reduces the value individuals place on their well-being".[28]
How Necessary Is It to Be ASSURED?
Many scientists cite ASSURED as a necessity and a large consensus for the ASSURED criteria among healthcare workers in, for instance, Uganda, has been found.[49] Pai et al. give another, more pragmatic and contextual viewpoint: ASSURED imposes artificial restrictions that may not be necessary, depending on the context in which the devices are used.[5] A device used for first line screening with the aim of referral to another, more specialized healthcare provider can have a lower specificity compared to a test that makes decisions about, or monitors, the treatment itself.
A study that compared 12 different combinations of hepatitis C virus-diagnostics, either PoC, lab-based, or a combination, found the cheapest strategy was a two-test combination, first using a lower-specificity PoC antibody test followed by a confirmation via an RNA PoC test. All one-step strategies showed higher false-positive rates and were not cost-effective under base-case assumptions.[50] Gift et al., reported as early as 1999 on the "rapid test paradox" and the connection between LTFU and PoC tests with limited sensitivity that lead to better treatment outcomes.[51]
Another aspect: only 28% of the inhabitants of Africa have access to advanced healthcare facilities. Tests that need minimal infrastructure could give an additional 47% of the African population access to diagnostic tests. While improving the accuracy for bacterial pneumonia tests in advanced healthcare facilities only led to 119,000 more disability-adjusted life-years (DALYs) saved per year, 263,000 more DALYs could be saved annually if this test would be made available for rural sites with minimal resources.[43] A PoC device that would require minimal laboratory infrastructure could prevent 138,000 congenital syphilis cases and 148,000 stillbirth per year; a PoC device that would not require any laboratory infrastructure at all would prevent 201,000 cases and 215,000 stillbirths.[22]
Another aspect criticized is that PoC tests often focus on a single disease; however, healthcare workers in LICs are concerned with syndromes of unknown etiology.[20] Having one test measure several factors would give the healthcare provider more diagnostic security, and save time.
Steps towards an Effectively Usable PoC Device
Multiplexing might be a large step forward, and nowadays several methods for multiplexing are possible and being developed.[4,22] Several authors suggest modern microfluidics to alleviate some of these problems. In sample preparation, for example, all the technology for DNA/RNA extraction and purification can be included in a small cartridge with very easy usability,[43] reducing the need for external equipment and thus reducing cost.[21] Many other authors agree and see great potential in microfluidics for LIC-PoC.[52–55] The widespread use of smartphones is considered in many publications as a possible readout-device for optical platforms, as well as an ICT-connector.[56–60]
To prepare nucleic assays for LICs environments, one major problem is assay stabilization, as PCR mixes require cold storage. Lyophillization would be an option to create assays that do not require a cold-chain. The problematic part is the diverse array of compounds used that are incompatible with freeze-drying, such as glycerol. There are mixes that are possible to freeze-dry; however, the amplification efficiency will likely suffer.[43] To improve ruggedness, functions should be reduced to the essential and have integrated quality control, and local production may help with access to support and consumables.[61]
The Market
Market Introduction
The step from prototype towards market introduction seems to be the most taxing step, as even low-cost PoC devices are being developed in many laboratories in HICs, but not being adopted as well in LICs.[28]
Funding and IP
Funding is not only a consideration of research, but also of valorization. Even when funding for novel research is available, additional funding for economic aspects, such as manufacturing, distribution and maintenance are harder to find, resulting in the abandonment of projects by the original researcher due to cost, as well as deterring investment and interest from companies, which favor secure, established products.[26] Bringing new medical devices to the market is a costly enterprise, developing a new drug exceeds USD 1 billion in cost, takes more than 10 years, and only 11.8–21% of new drugs are approved. There are no discrete numbers for medical devices, given that the field is highly diverse with an estimate of over 500,000 different types of devices, which span from X-Ray machines to hip implants with different diverse aspects.[62] These processes can take years until a patient reaps the benefit of a new product, if it reaches the market after all.[63] This might make it unprofitable for a company to conduct fundamental research for LICs PoC if there is not an immediate benefit in reach.[3]
Next to funding, intellectual property (IP) proves to be a large barrier for the development of systems upon existing technologies. Especially IP on molecules and genes make this increasingly difficult.[4] Existing patents covering biomarkers and even entire organisms exist, and there are large IP barriers in diagnostic platforms and/or components of diagnostic platforms.[21,22] One solution for IP considerations are new private–public partnerships, such as the World Intellectual Property Organization (WIPO) Re:search consortium. WIPO is the UN agency tasked with developing international IP systems fostering innovation for NTDs that benefits everyone. WIPO Re:search enables members royalty free use of infrastructure, compound libraries, IP assets and know-how. The WIPO consists of 107 members in 30 countries.
Regulations
High regulatory barriers and strict healthcare standards are also a significant barrier to the introduction of innovations.[4,5,20] While good regulations are important, they are difficult and require substantial expertise to navigate them, which discourages innovation, especially in LICs where profitable returns are unsure. Clear and straightforward national policies for diagnostic evaluation and certification are key.[20] The pharmaceutical world is more advanced in setting up harmonization infrastructure, which is still lagging behind for diagnostics. In total, 23 countries in Africa banded together and pledged to harmonize the approval method for diagnostics, which is a huge step, as companies do not have to acquire approval for each country separately.[66] A WHO program that examines the quality and the safety of HIV and Malaria tests, "prequalification", is a helpful guide for LICs, helping governments to speed up the approval process.[26]
Integrated Market Expertise
Missing collaboration between academia and industry is another bottleneck. A multidisciplinary team for market introduction is an important part of the valorization trajectory.[4,21] A study of 358 medical devices for LICs found only 134 met the study requirements to count as "commercialized".[61] Of the hundreds of devices beyond that study, many likely failed to commercialize because of a failed transition from prototype to market introduction.[61] Those aspects of quality control and assurance and supply chain management are what companies have strong expertise in.
For this, it is not only necessary to have a research and valorization team, but also specialists for regulation, culture and policy.[21] Especially researchers in low-income-countries often lack the knowledge spanning all involved fields, from discovery and research to market introduction.[3] The device must consider local and regional constraints, involved stakeholders and their needs and the capacity of the local healthcare workforce, but also social and cultural contexts for things that can easily be overlooked, e.g., whether blood sampling is easily accepted.[5,22,61] One target group that should be especially focused on, but are often not included, are the end-users, who should be integrated in each step of the design process.[20,28,45]
Market Penetration
Device Quality
After the introduction of the product, other aspects become important for wider market penetration. One problem is bad batch-to-batch reproducibility especially in lateral flow devices that hamper upscaling.[21] Low device quality and low reproducibility can lead to decreased trust by healthcare providers and thus affect the adoption of other devices negatively.[28] This is one of the greatest concerns of rural LICs healthcare practitioners. Another study reported doubts from healthcare practitioners regarding test trustworthiness with either the accuracy, the robustness or the clarity of results. Adding to distrust is the concern of counterfeit tests being delivered.[49]
Economic and Social Placement
Even a theoretical PoC device that achieved all ASSURED criteria perfectly might not be sustainable and gain market acceptance if they do not have a viable business model attached to them.[45] The large importance of proper economic placement is shown by the fact that even inaccurate devices can be a market success if the stakeholders along the value chain profit from their use. For example, inaccurate serological tuberculosis tests, which the WHO advises against, are very common in private healthcare facilities in India, as well as in 17 of the top 22 other countries most affected by the disease. Private doctors earn referral money and other incentives for ordered tests, leading to an overreliance on inaccurate diagnostics due to economic incentives.[5,67]
Other examples of misplacement can be found in India and South Africa, where many well-working and cheap (USD 1 per test) PoC tests are available for different diseases, such as HIV, Malaria, Dengue, Syphilis and Hepatitis. Yet many of those tests are still not commonly used at home, the physician's office or even in rural healthcare clinics. Instead, testing happens foremost in laboratories and hospitals, whereas small independent laboratories are the major users of PoC devices.[5] Lab personnel are often skeptical against testing outside of controlled settings, as they lose control over testing and quality assurance and it interferes with their business model.[20,45]
Market penetration is also heavily dependent on price. A guideline to evaluate the affordability is 1–3× the GDP per capita per quality adjusted life years, which is gained through the intervention.[50] However, this calculation is not commonly shared as a good estimate and it may not lead to the best investment. Patients themselves may not be willing to pay the suggested price as they lack money. In Kenya, 51% of healthcare expenses are payed out of pocket, healthcare costs are often covered through "harambees" fundraising events in the community; 46% of the population only has USD 1 or less to spend each day.[28] A good example for this is HIV diagnosis and monitoring. Since viral counts are difficult to do in the field, often a laboratory procedure is required. While PoC HCTs devices are established on the market, widespread use is limited by the initial high cost of the device. Research showed it can be viable for clinics with a moderate or large amount of patients to have such PoC HCTs devices for HIV detection, as the initial high cost can be distributed over more patients.[71]
While many simulations of cost effectiveness base their assumption on high-prevalence areas, others remark that especially for "the last mile", in areas with lack of infrastructure, PoC might be the only viable alternative for the hardest to reach 10% of patients, as transport networks get more and more difficult to establish in remote areas.[75]
Product Distribution
Limited infrastructure in LICs not only results in low return-of-investments for companies, but also makes the distribution of the device and technical support more difficult, which might discourage companies or hamper the market penetration.[26] Stock outs and supply network problems are a massive obstacle to market penetration. In a scale-up program for syphilis tests from an NGO-led pilot to a ministry of health operated large scale operation in Zambia, half of the pilot sites suffered at least one stock out. PoC for pregnant women also reported stock outs in several stages of the study, with up to 60% of sites reporting stock outs. The longest time the device was out of stock was a median of 6 weeks. In Uganda, malaria diagnostic tests were only available in 24% of 125 lower healthcare facilities, and 72% of community healthcare workers did not receive malaria testing kits for 6 month.[2]
An innovative solution for the fast distribution of medical products in LICs is shown by the company Zipline, which uses remote drones to distribute blood preservations to hospitals in need all over Rwanda. Replacing the delivery from taking hours by Motorbike, to mere minutes by a Zipline drone, this innovation reduces the number of blood stocks the hospital needs and thus reduces waste due to expiry. In emergencies, matching blood can be delivered within minutes, something impossible with motorcycle rides that could take up to 5 hours.[80,81] In the recent COVID-19 pandemic, Zipline collected COVID-19 test-samples by car and sent them bundled together to large hospitals via their drones, while also distributing to hospitals other COVID-19 related necessities.[82,83]
The Usage
Limited testing capabilities are often a bottleneck for adequate therapy. HIV treatment, where CD4+ counts and viral load are used to monitor antiretroviral therapy (ART), is a prime example. In Sub-Saharan Africa, the median of patients retained between HIV diagnosis and CD4+ count was 59%.[85]
Healthcare Management
Political will towards PoC might be reduced when PoC tests lead to more demand for treatment, while at the same time treatment capabilities are scarce.[5] When treatment is available, patients might also just opt-out of tests in favor of direct use of medication, such as over-the-counter-antibiotics. This has been reported in Thailand, where missing information about disease origin among the public leads to a preference of medication instead of proper diagnosis.[92]
Information technology plays a key role in the context of PoC. The rapid reporting of results and counseling via mobile phones are essential for a decentralized use of PoC. The usage of mobile phones in this way is generally categorized as mHealth, and concerns itself with the use of wireless technology instead of connection through ordinary landline infrastructure, such as in eHealth. This is especially interesting for LMICs, as mobile phone usage outperforms other communication infrastructure usage.[98] In total, 70% of the 7.4 billion users of cellular phones reside in LMICs, and especially in sub-Saharan Africa, mHealth had a rapid expansion, making this approach hugely promising.[20,99,100]
Training
While large hospitals in central areas might also have an appropriate workforce, the staff in rural healthcare clinics, which are the main access points to healthcare for the rural population, consist mainly of untrained individuals; often just one doctor, nurse or pharmacist, with the possible addition of lay healthcare workers (LHWs).[43,61,95,101]
Human resources are in surprisingly short supply when it comes to healthcare workers and may stretch out the system. There is also a problem in the lack of educated healthcare personnel, especially in Africa, which has over 24% of the worldwide disease burden while only having 2% of physicians of the world.[61,103–105]
Surveyed healthcare workers assessed PoC diagnostics as easy to use; however, they still expressed fear of knowledge gaps among their users and concern of incorrect use. For example, the use of a wrong buffer solution or no buffer solution at all was observed.[49] Despite those fears, the world health organization recommends task shifting to LHWs to meet human resources needs.[78,85]
The question is whether task shifting can be achieved without a loss in reliability. A study about task shifting for the use of the HCTs Pima CD4+ Analyzer (Allere) in Namibia showed that lay-health-workers can produce valid tests as nurses. In a large study of 1429 CD4+ tests, in which 500 were performed by nurses and 929 by LHWs, the reception of test results by the patients was in favor of LHWs, with 98.1% contrary to 95.6%. LHCs were only slightly slower, with a median turnaround time of 21 minutes compared to 20 minutes for nurses, both a tremendous improvement from the median laboratory turnaround time of 4 days.[85]
Use by Trained Doctors
In cases where trained medical professionals are available, the bottlenecks present themselves differently. In India, doctors prefer clinical diagnoses coupled with empiric treatments over a higher diagnostic security. Broadband antibiotic prescription after only a short symptomatic observation is a common example.[5]
View on PoC
With regard to the view of patients on PoC, 97% were satisfied with PoC results and 96% would recommend them. However, only half of clinicians thought they would give reliably accurate results; 46% were unsure and 4% considered them not accurate. In total, 65% of healthcare workers used medication even on a negative test, showing that trust in the test is limited.[28]
Conclusions
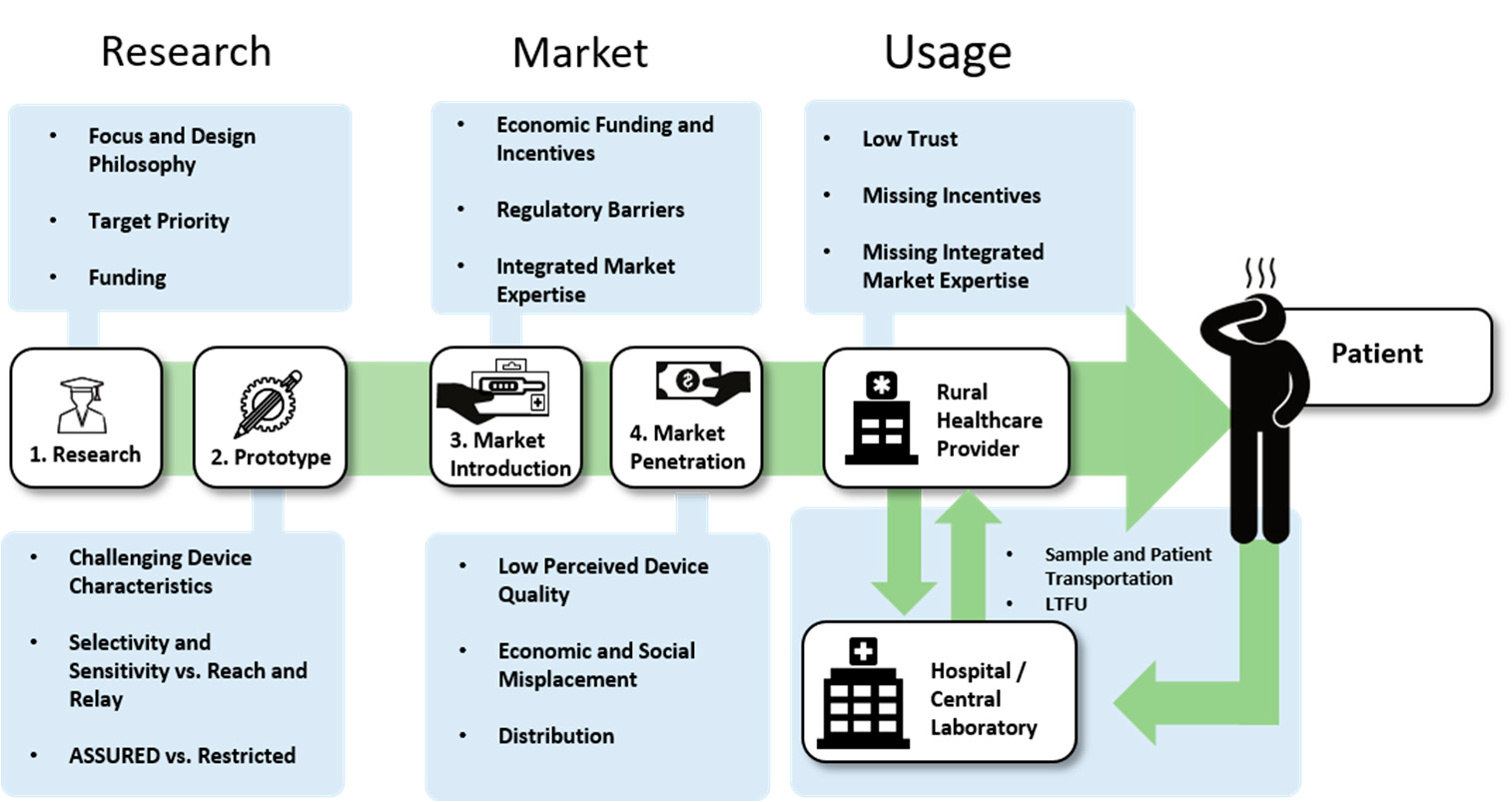
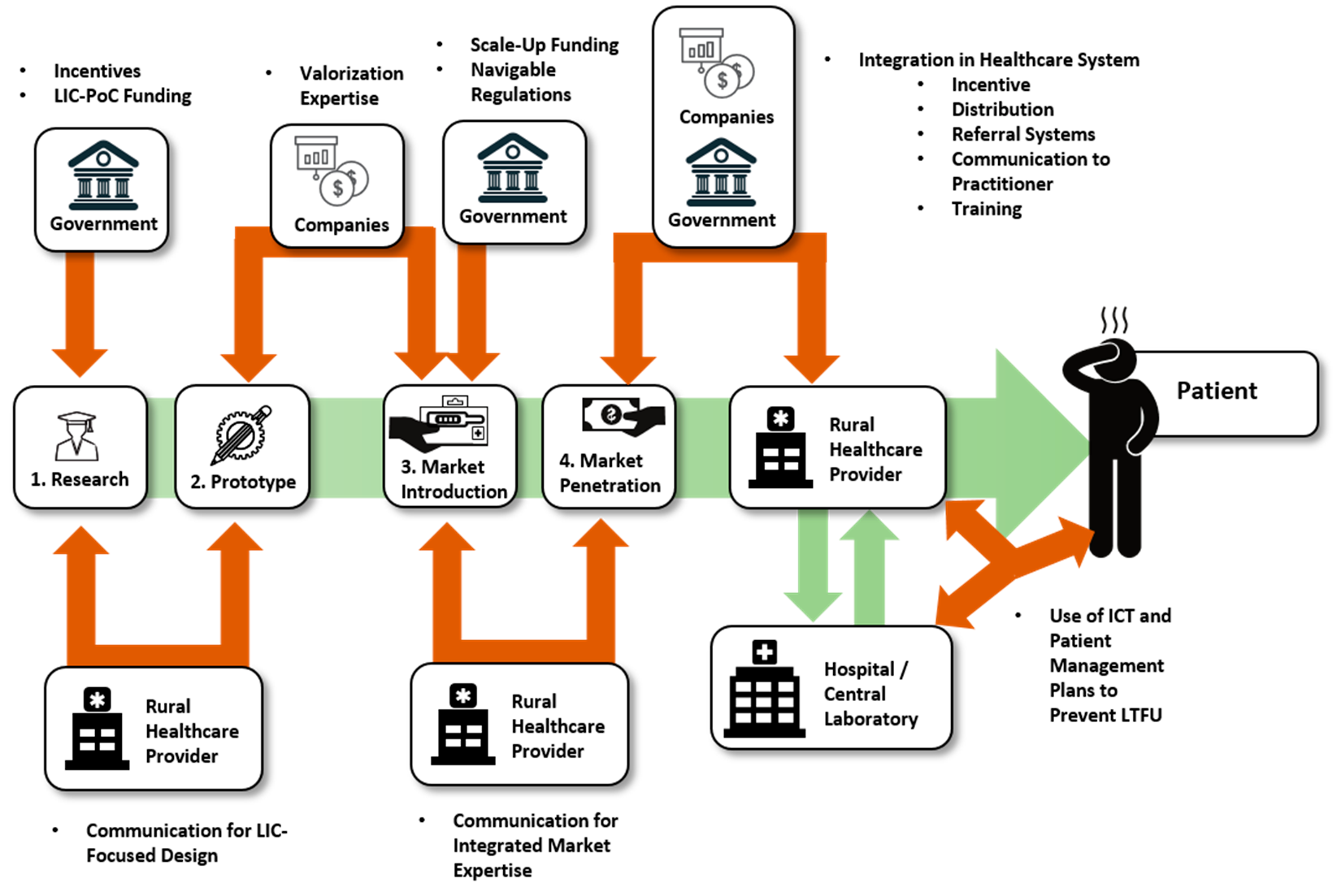
Fundamental research always starts with funding, and it is an obvious consideration. However, funding is not only needed here. Additional funding, as well as incentives for valorization is something direly needed to actually make the jump from a research principle to a medical device. Push- and pull-incentives are used with considerable success in drug development and might prove valuable if systems directed at PoC are in place. Connecting all stakeholders, research groups, companies, healthcare professionals, governments and NGOs, is essential.
For the development of the device, one can ask how important it is to be ASSURED. While many argue for sensitivity, specificity and reliability as a main point, there are other voices arguing for a more integrated view. A two-step system can be cheaper and more specific, despite the first stage not having optimal characteristics. However, for this to work, a proper integration into the healthcare system, with reasonable referral structures and minimal LTFU, has to be achieved.
On the market side, the tremendous influence of a proper placement into the healthcare system and its incentive structure is shown by the use of subpar-PoC devices in India and other countries. If malfunctioning PoC devices can achieve widespread use, surely working diagnostics can be used if they are properly integrated into an incentive structure. A social problem identified as a large bottleneck is the lack of available workforce for testing. While the opinion of researchers regarding task shifting varies from skeptical to enthusiastic, there are interesting insights into its feasibility, arguing that lay-healthcare-workers can even conduct tests on more difficult platforms if trained properly.
As research on PoC diagnostics continues, we will get closer to versatile, accurate and cheap detection methods that are more in line with the desirable ASSURED criteria. However, for the immediate success of PoC and for the benefit of patients in LICs, the research part might already be more advanced compared to the other aspects of the value chain. A wide range of reasons can be identified, from missing funding for scale-up and lack of corporate incentives, as well as delivery problems and stock outs, to the problem of integration in healthcare systems and lacking trust by doctors. The lack of valorization of PoC in LICs seems to be a social and an economical problem, more than a problem of research.
While Zipline is a company concerned with infrastructure and not with diagnostics, it still shows, in an impressive manner, how new technology can innovate a whole market, if there is political will and no old stakeholders that benefit from the status quo. Zipline's CEO, Keller Rinaudo, stated that the technology is the easy part, it is more difficult to improve regulatory issues, acquiring and training the necessary workforce locally and creating awareness of their services to doctors and healthcare workers.
Faced with the healthcare challenges in LICs, a transformation to a smart healthcare system, with real-time information flow and referral structures, will be necessary to make the most out of the innovations that come out of the lab. There is a case to be made that scientific progress and innovation may not be the limiting factor, and other limiting steps seem to be hindering valorization at least as much. The research and implementation of a new device have to be designed in synergy with its target location, instead of merely adapting to it later on. Research needs to break down as many barriers and enable the connection of as many stakeholders as possible in order for policy makers and companies to take the leap and bring PoC diagnostics to the patients.
References
- Boutayeb, A. The impact of infectious diseases on the development of Africa. In The Handbook of Disease Burdens and Quality of Life Measures; Preedy, V.R., Watson, R.R., Eds.; Springer: New York, NY, USA, 2010; pp. 1171–1188.
- Kuupiel, D.; Bawontuo, V.; Drain, P.K.; Gwala, N.; Mashamba-Thompson, T.P. Supply chain management and accessibility to point-of-care testing in resource-limited settings: A systematic scoping review. BMC Heal. Serv. Res. 2019, 19, 519.
- Starr, A.; Graef, K.M.; Dent, J. Fostering innovative product development for neglected tropical diseases through partnerships. Pharm. Pat. Anal. 2016, 5, 391–400.
- Migliozzi, D.; Guibentif, T. Assessing the potential deployment of biosensors for point-of-care diagnostics in developing countries: Technological, economic and regulatory aspects. Biosensors 2018, 8, 119.
- Pai, N.P.; Vadnais, C.; Denkinger, C.; Engel, N.; Pai, M. Point-of-care testing for infectious diseases: Diversity, complexity, and barriers in low-and middle-income countries. PLoS Med. 2012, 9, e1001306.
- Sia, S.K.; Kricka, L.J. Microfluidics and point-of-care testing. Lab Chip 2008, 8, 1982–1983.
- Kozel, T.R.; Burnham-Marusich, A.R. Point-of-care testing for infectious diseases: Past, present, and future. J. Clin. Microbiol. 2017, 55, 2313–2320.
- Pashchenko, O.; Shelby, T.; Banerjee, T.; Santra, S.A. Comparison of optical, electrochemical, magnetic, and colorimetric point-of-care biosensors for infectious disease diagnosis. ACS Infect. Dis. 2018, 4, 1162–1178.
- Lowdon, J.W.; Eersels, K.; Rogosic, R.; Boonen, T.; Heidt, B.; Diliën, H.; Van Grinsven, B.; Cleij, T.J. Surface grafted molecularly imprinted polymeric receptor layers for thermal detection of the new psychoactive substance 2-methoxphenidine. Sens. Actuators 2019, 295, 586–595.
- Vandenryt, T.; Van Grinsven, B.; Eersels, K.; Cornelis, P.; Kholwadia, S.; Cleij, T.J.; Thoelen, R.; De Ceuninck, W.; Peeters, M.; Wagner, P. Single-shot detection of neurotransmitters in whole-blood samples by means of the heat-transfer method in combination with synthetic receptors. Sensors 2017, 17, 2701.
- Takemura, K.; Adegoke, O.; Takahashi, N.; Kato, T.; Li, T.-C.; Kitamoto, N.; Tanaka, T.; Suzuki, T.; Park, E.Y. Versatility of a localized surface plasmon resonance-based gold nanoparticle-alloyed quantum dot nanobiosensor for immunofluorescence detection of viruses. Biosens. Bioelectron. 2017, 89, 998–1005.
- Lowdon, J.W.; Eersels, K.; Rogosic, R.; Heidt, B.; Diliën, H.; Redeker, E.S.; Peeters, M.; Van Grinsven, B.; Cleij, T.J. Substrate displacement colorimetry for the detection of diarylethylamines. Sens. Actuators 2019, 282, 137–144.
- Xiong, L.-H.; Cui, R.; Zhang, Z.-L.; Yu, X.; Xie, Z.; Shi, Y.-B.; Pang, D.-W. Uniform fluorescent nanobioprobes for pathogen detection. ACS Nano 2014, 8, 5116–5124.
- Myers, F.B.; Lee, L.P. Innovations in optical microfluidic technologies for point-of-care diagnostics. Lab Chip 2008, 8, 2015–2031.
- Afsahi, S.; Lerner, M.B.; Goldstein, J.M.; Lee, J.; Tang, X.; Bagarozzi, D.A.; Pan, D.; Locascio, L.; Walker, A.; Barron, F.E.; et al. Novel graphene-based biosensor for early detection of Zika virus infection. Biosens. Bioelectron. 2018, 100, 85–88.
- Campuzano, S.; Yáñez-Sedeño, P.; Pingarrón, J.M. Molecular biosensors for electrochemical detection of infectious pathogens in liquid biopsies: Current trends and challenges. Sensors 2017, 17, 2533.
- Cecchetto, J.; Fernandes, F.C.B.; Lopes, R.; Bueno, P.R. The capacitive sensing of NS1 Flavivirus biomarker. Biosens. Bioelectron. 2017, 87, 949–956.
- Hsieh, K.; Ferguson, B.S.; Eisenstein, M.; Plaxco, K.W.; Soh, H.T. Integrated electrochemical microsystems for genetic detection of pathogens at the point of care. Acc. Chem. Res. 2015, 48, 911–920.
- Park, J.Y.; Kricka, L.J. Prospects for nano-and microtechnologies in clinical point-of-care testing. Lab Chip 2007, 7, 547–549.
- Kuupiel, D.; Bawontuo, V.; Mashamba-Thompson, T.P. Improving the accessibility and efficiency of point-of-care diagnostics services in low- and middle-income countries: Lean and agile supply chain management. Diagnostics 2017, 7, 58.
- Wang, S.; Lifson, M.A.; Inci, F.; Liang, L.-G.; Sheng, Y.-F.; Demirci, U. Advances in addressing technical challenges of point-of-care diagnostics in resource-limited settings. Expert Rev. Mol. Diagn. 2016, 16, 449–459.
- Urdea, M.; Penny, L.A.; Olmsted, S.S.; Giovanni, M.Y.; Kaspar, P.; Shepherd, A.; Wilson, P.; Dahl, C.A.; Buchsbaum, S.; Moeller, G.; et al. Requirements for high impact diagnostics in the developing world. Nature 2006, 444 (Suppl. 1), 73–79.
- Tayoun, A.N.A.; Ward, B.P.; Maltezos, G.; Scherer, A.; Tsongalis, G.J. Evaluating the thermostability of commercial fast real-time PCR master mixes. Exp. Mol. Pathol. 2012, 93, 261–263.
- Furuse, Y. Analysis of research intensity on infectious disease by disease burden reveals which infectious diseases are neglected by researchers. Proc. Natl. Acad. Sci. USA 2019, 116, 478–483.
- Elliott, A.M.; Luo, N.; Tembo, G.; Halwiindi, B.; Steenbergen, G.; Machiels, L.; Pobee, J.; Nunn, P.; Hayes, R.; McAdam, K.P. Impact of HIV on tuberculosis in Zambia: A cross sectional study. BMJ 1990, 301, 412–415.
- McNerney, R. Diagnostics for developing countries. Diagnostics 2015, 5, 200–209.
- Hecht, R.; Stover, J.; Bollinger, L.; Muhib, F.; Case, K.K.; De Ferranti, D. Financing of HIV/AIDS programme scale-up in low-income and middle-income countries, 2009–2031. Lancet 2010, 376, 1254–1260.
- Kimani, F.W.; Mwangi, S.M.; Kwasa, B.; Kusow, A.M.; Ngugi, B.; Chen, J.; Liu, X.; Cademartiri, R.; Thuo, M.M. Rethinking the design of low-cost point-of-care diagnostic devices. Micromachines 2017, 8, 317.
- Expedited Programs for Serious Conditions, Drugs and Biologics. Available online: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/expedited-programs-serious-conditions-drugs-and-biologics (accessed on 9 July 2020).
- Ridley, D.B.; Régnier, S.A. The commercial market for priority review vouchers. Health Aff. 2016, 35, 776–783.
- Slingsby, B.; Kurokawa, K. The Global Health Innovative Technology (GHIT) Fund: Financing medical innovations for neglected populations. Lancet Glob. Health 2013, 1, e184–e185.
- Bessa, T.C.B.; De Aragão, E.S.; Guimarães, J.M.M.; Almeida, B.D.A. R&D in vaccines targeting neglected diseases: An exploratory case study considering funding for preventive tuberculosis vaccine development from 2007 to 2014. Biomed Res. Int. 2017, 2017, 4765719.
- Mercer, M.A.; Thompson, S.M.; De Araujo, R.M. The role of international NGOs in health systems strengthening: The case of Timor-Leste. Int. J. Health Serv. 2014, 44, 323–335.
- Black, R.E.; Bhan, M.K.; Chopra, M.; Rudan, I.; Victora, C.G. Accelerating the health impact of the Gates Foundation. Lancet 2009, 373, 1584–1585.
- Guha-Sapir, D. The Gates Foundation: Looking at the bigger picture. Lancet 2009, 374, 201–202.
- McCoy, D.C.; Kembhavi, G.; Patel, J.; Luintel, A. The Bill & Melinda Gates Foundation's grant-making programme for global health. Lancet 2009, 373, 1645–1653.
- Rao, B.C. The science underlying frugal innovations should not be frugal. R. Soc. Open Sci. 2019, 6, 180421.
- Prabhu, J. Frugal innovation: Doing more with less for more. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2017, 375, 20160372.
- Tran, V.-T.; Ravaud, P. Frugal innovation in medicine for low resource settings. BMC Med. 2016, 14, 102.
- Howitt, P.; Darzi, A.; Yang, G.-Z.; Ashrafian, H.; Atun, R.; Barlow, J.; Blakemore, A.; Bull, A.M.J.; Car, J.; Conteh, L.; et al. Technologies for global health. Lancet 2012, 380, 507–535.
- Heidt, B.; Rogosic, R.; Bonni, S.; Jansen, J.P.; Dimech, D.; Lowdon, J.W.; Arreguin-Campos, R.; Redeker, E.S.; Eersels, K.; Diliën, H.; et al. The liberalization of microfluidics: Form 2 benchtop 3D printing as an affordable alternative to established manufacturing methods. Phys. Status Solidi (A) 2020.
- Kleinman, Z. Coronavirus: 3D Printers Save Hospital with Valves. BBC News [Online], 16 March 2020.
- Tayoun, A.N.A.; Burchard, P.R.; Malik, I.; Scherer, A.; Tsongalis, G.J. Democratizing molecular diagnostics for the developing world. Am. J. Clin. Pathol. 2014, 141, 17–24.
- Zachariah, R.; Reid, S.D.; Chaillet, P.; Massaquoi, M.; Schouten, E.J.; Harries, A.D. Viewpoint: Why do we need a point-of-care CD4 test for low-income countries? Trop. Med. Int. Health 2011, 16, 37–41.
- Mashamba-Thompson, T.P.; Jama, N.A.; Sartorius, B.; Drain, P.K.; Thompson, R.M. Implementation of point-of-care diagnostics in rural primary healthcare clinics in South Africa: Perspectives of key stakeholders. Diagnostics 2017, 7, 3.
- Toskin, I.; Murtagh, M.; Peeling, R.W.; Blondeel, K.; Cordero, J.P.; Kiarie, J. Advancing prevention of sexually transmitted infections through point-of-care testing: Target product profiles and landscape analysis. Sex. Transm. Infect. 2017, 93, S69–S80.
- Peeling, R.W.; Holmes, K.K.; Mabey, D. Rapid tests for sexually transmitted infections (STIs): The way forward. Sex. Transm. Infect. 2006, 82, v1–v6.
- Peeling, R.W.; Mabey, D.; Herring, A.; Hook, E.W. Why do we need quality-assured diagnostic tests for sexually transmitted infections? Nat. Rev. Genet. 2006, 4, S7–S19.
- Rasti, R.; Nanjebe, D.; Karlström, J.; Muchunguzi, C.; Mwanga-Amumpaire, J.; Gantelius, J.; Mårtensson, A.; Rivas, L.; Galban, F.; Reuterswärd, P.; et al. Health care workers' perceptions of point-of-care testing in a low-income country: A qualitative study in Southwestern Uganda. PLoS ONE 2017, 12, e0182005.
- Duchesne, L.; Hejblum, G.; Kane, N.C.T.; Njouom, R.; Toni, T.-D.; Moh, R.; Sylla, B.; Rouveau, N.; Attia, A.; Lacombe, K. Model-based cost-effectiveness estimates of testing strategies for diagnosing hepatitis C virus infection in people who use injecting drugs in Senegal. Int. J. Drug Policy 2020, 75, 102613.
- Gift, T.L.; Pate, M.S.; Hook, E.W.; Kassler, W.J. The rapid test paradox: When fewer cases detected lead to more cases treated: A decision analysis of tests for Chlamydia trachomatis. Sex. Transm. Dis. 1999, 26, 232–240.
- Pandey, C.M.; Augustine, S.; Kumar, S.; Kumar, S.; Nara, S.; Srivastava, S.; Malhotra, B.D. Microfluidics based point-of-care diagnostics. Biotechnol. J. 2017, 13, 1700047.
- Azhar, M.; Dendukuri, D. Microfluidic platforms for point of care (POC) medical diagnostics. In Medical Biosensors for Point of Care (POC) Applications; Narayan, R., Ed.; Woodhead Publishing is an Imprint of Elsevier: Duxford, UK, 2016; pp. 255–273.
- Chin, C.D.; Linder, V.; Sia, S.K. Commercialization of microfluidic point-of-care diagnostic devices. Lab. Chip. 2012, 12, 2118–2134.
- Sharma, S.; Zapatero-Rodríguez, J.; Estrela, P.; O'Kennedy, R. Point-of-care diagnostics in low resource settings: Present status and future role of microfluidics. Biosensors 2015, 5, 577–601.
- Zhdanov, A.; Keefe, J.; Franco-Waite, L.; Konnaiyan, K.R.; Pyayt, A. Mobile phone based ELISA (MELISA). Biosens. Bioelectron. 2018, 103, 138–142.
- Archibong, E.; Konnaiyan, K.R.; Kaplan, H.; Pyayt, A. A mobile phone-based approach to detection of hemolysis. Biosens. Bioelectron. 2017, 88, 204–209.
- Quesada-González, D.; Merkoçi, A. Mobile phone-based biosensing: An emerging "diagnostic and communication" technology. Biosens. Bioelectron. 2017, 92, 549–562.
- Sun, A.C.; Yao, C.; Venkatesh, A.G.; Hall, D.A. An efficient power harvesting mobile phone-based electrochemical biosensor for point-of-care health monitoring. Sens. Actuators 2016, 235, 126–135.
- Delaney, J.L.; Hogan, C.F. Mobile phone based electrochemiluminescence detection in paper-based microfluidic sensors. Methods Mol. Biol. 2015, 1256, 277–289.
- Sarvestani, A.S.; Sienko, K. Medical device landscape for communicable and noncommunicable diseases in low-income countries. Glob. Health 2018, 14, 65.
- Parvizi, N.; Woods, K. Regulation of medicines and medical devices: Contrasts and similarities. Clin. Med. 2014, 14, 6–12.
- Basu, S.; Hassenplug, J.C. Patient access to medical devices-A comparison of US and European review processes. N. Engl. J. Med. 2012, 367, 485–488.
- Ramamoorthi, R.; Graef, K.M.; Dent, J. WIPO Re:Search: Accelerating anthelmintic development through cross-sector partnerships. Int. J. Parasitol. Drugs Drug Resist. 2014, 4, 220–225.
- Manner, C.K.; Graef, K.M.; Dent, J. WIPO Re:Search: Catalyzing Public-Private Partnerships to Accelerate Tropical Disease Drug Discovery and Development. Trop. Med. Infect. Dis. 2019, 4, 53.
- Peeling, R. Bringing diagnostics to developing countries: An interview with Rosanna Peeling. Expert Rev. Mol. Diagn. 2015, 15, 1107–1110.
- Jaroslawski, S.; Pai, M. Why are inaccurate tuberculosis serological tests widely used in the Indian private healthcare sector? A root-cause analysis. J. Epidemiol. Glob. Health 2012, 2, 39–50.
- Baloch, N.A.; Pai, M. Tuberculosis control: Business models for the private sector. Lancet Infect. Dis. 2012, 12, 579–580.
- Marseille, E.; Larson, B.A.; Kazi, D.S.; Kahn, J.G.; Rosen, S.B. Thresholds for the cost–effectiveness of interventions: Alternative approaches. Bull. World Health Organ. 2015, 93, 118–124.
- Campos, N.G.; Tsu, V.D.; Jeronimo, J.; Mvundura, M.; Kim, J.J. Estimating the value of point-of-care HPV testing in three low-and middle-income countries: A modeling study. BMC Cancer 2017, 17, 791.
- Simeon, K.; Sharma, M.; Dorward, J.; Naidoo, J.; Dlamini, N.; Moodley, P.; Samsunder, N.; Barnabas, R.V.; Garrett, N.; Drain, P.K. Comparative cost analysis of point-of-care versus laboratory-based testing to initiate and monitor HIV treatment in South Africa. PLoS ONE 2019, 14, e0223669.
- Phillips, A.; Cambiano, V.; Nakagawa, F.; Ford, D.; Apollo, T.; Murungu, J.; Rousseau, C.; Garnett, G.; Ehrenkranz, P.; Bansi-Matharu, L.; et al. Point-of-care viral load testing for Sub-Saharan Africa: Informing a target product profile. Open Forum Infect. Dis. 2016, 3, ofw161.
- Hyle, E.P.; Jani, I.; Lehe, J.; Su, A.E.; Wood, R.; Quevedo, J.; Losina, E.; Bassett, I.V.; Pei, P.P.; Paltiel, A.D.; et al. The clinical and economic impact of point-of-care CD4 testing in Mozambique and other resource-limited settings: A cost-effectiveness analysis. PLoS Med. 2014, 11, e1001725.
- Estill, J.; Egger, M.; Blaser, N.; Salazar-Vizcaya, L.; Garone, D.; Wood, R.; Campbell, J.; Hallett, T.B.; Keiser, O.; Africa, I.S. Cost-effectiveness of point-of-care viral load monitoring of antiretroviral therapy in resource-limited settings: Mathematical modelling study. AIDS 2013, 27, 1483–1492.
- Nichols, B.E.; Girdwood, S.J.; Crompton, T.; Stewart-Isherwood, L.; Berrie, L.; Chimhamhiwa, D.; Moyo, C.; Kuehnle, J.; Stevens, W.; Rosen, S.; et al. Monitoring viral load for the last mile: What will it cost? J. Int. Aids Soc. 2019, 22, e25337.
- Girdwood, S.J.; Nichols, B.E.; Moyo, C.; Crompton, T.; Chimhamhiwa, D.; Rosen, S. Optimizing viral load testing access for the last mile: Geospatial cost model for point of care instrument placement. PLoS ONE 2019, 14, e0221586.
- Smith, A.; Sabidó, M.; Camey, E.; Batres, A.; Casabona, J. Lessons learned from integrating simultaneous triple point-of-care screening for syphilis, hepatitis B, and HIV in prenatal services through rural outreach teams in Guatemala. Int. J. Gynecol. Obstet. 2015, 130, S70–S72.
- Yao, K.; Wafula, W.; Bile, E.; Cheignsong, R.; Howard, S.; Demby, A.; Nkengasong, J. Ensuring the quality of HIV rapid testing in resource-poor countries using a systematic approach to training. Am. J. Clin. Pathol. 2010, 134, 568–572.
- Engel, N.; Wachter, K.; Pai, M.; Gallarda, J.; Boehme, C.; Celentano, I.; Weintraub, R. Addressing the challenges of diagnostics demand and supply: Insights from an online global health discussion platform. BMJ Glob. Health 2016, 1, e000132.
- Ackerman, E.; Strickland, E. Medical delivery drones take flight in East Africa. IEEE Spectr. 2018, 55, 34–35.
- Ackerman, E.; Koziol, M. The blood is here: Zipline's medical delivery drones are changing the game in Rwanda. IEEE Spectr. 2019, 56, 24–31.
- Rustici, C. Drone Start-up Zipline is Helping Fight Covid-19 in Africa. MedicalExpo e-Magazine [Online], 13 June 2020.
- Lewis, N. Zipline is Delivering COVID-19 Medical Supplies to Rural Ghana and Rwanda by Drone. Business Insider [Online], 12 May 2020.
- Scott, J.E. Drone delivery models for healthcare. In Proceedings of the 50th Hawaii International Conference on System Sciences (HICSS-50), Hilton Waikoloa Village, Hawaii, HI, USA, 4–7 January 2017.
- Kaindjee-Tjituka, F.; Sawadogo, S.; Mutandi, G.; Maher, A.D.; Salomo, N.; Mbapaha, C.; Neo, M.; Beukes, A.; Gweshe, J.; Muadinohamba, A.; et al. Task-shifting point-of-care CD4+ testing to lay health workers in HIV care and treatment services in Namibia. Afr. J. Lab. Med. 2017, 6, 643.
- Hsiao, N.-Y.; Stinson, K.; Myer, L. Linkage of HIV-infected infants from diagnosis to antiretroviral therapy services across the Western Cape, South Africa. PLoS ONE 2013, 8, e55308.
- Ciaranello, A.L.; Park, J.-E.; Ramirez-Avila, L.; Freedberg, K.A.; Walensky, R.P.; Leroy, V. Early infant HIV-1 diagnosis programs in resource-limited settings: Opportunities for improved outcomes and more cost-effective interventions. BMC Med. 2011, 9, 59.
- Dunning, L.; Hsiao, N.-Y.; Myer, L. Point-of-care HIV early infant diagnosis: Is test sensitivity everything? J. Int. Aids Soc. 2015, 18, 20235.
- Ndlovu, Z.; Fajardo, E.; Mbofana, E.; Maparo, T.; Garone, D.; Metcalf, C.; Bygrave, H.; Kao, K.; Zinyowera, S. Multidisease testing for HIV and TB using the GeneXpert platform: A feasibility study in rural Zimbabwe. PLoS ONE 2018, 13, e0193577.
- Zeh, C.; Rose, C.E.; Inzaule, S.; Desai, M.A.; Otieno, F.; Humwa, F.; Akoth, B.; Omolo, P.; Chen, R.T.; Kebede, Y.; et al. Laboratory-based performance evaluation of PIMA CD4+ T-lymphocyte count point-of-care by lay-counselors in Kenya. J. Immunol. Methods 2017, 448, 44–50.
- Hsiao, N.-Y.; Dunning, L.; Kroon, M.; Myer, L. Laboratory evaluation of the alere q point-of-care system for early infant HIV diagnosis. PLoS ONE 2016, 11, e0152672.
- Haenssgen, M.J.; Charoenboon, N.; Althaus, T.; Greer, R.; Intralawan, D.; Lubell, Y. The social role of C-reactive protein point-of-care testing to guide antibiotic prescription in Northern Thailand. Soc. Sci. Med. 2018, 202, 1–12.
- Gething, P.W.; Johnson, F.A.; Frempong-Ainguah, F.; Nyarko, P.; Baschieri, A.; Aboagye, P.; Falkingham, J.; Matthews, Z.; Atkinson, P.M. Geographical access to care at birth in Ghana: A barrier to safe motherhood. BMC Public Health 2012, 12, 991.
- Tayler-Smith, K.; Zachariah, R.; Massaquoi, M.; Manzi, M.; Pasulani, O.; Akker, T.V.D.; Bemelmans, M.; Bauernfeind, A.; Mwagomba, B.; Harries, A.D.D. Unacceptable attrition among WHO stages 1 and 2 patients in a hospital-based setting in rural Malawi: Can we retain such patients within the general health system? Trans. R. Soc. Trop. Med. Hyg. 2010, 104, 313–319.
- Reid, S.; Fidler, S.J.; Cooke, G.S. Tracking the progress of HIV: The impact of point-of-care tests on antiretroviral therapy. Clin. Epidemiol. 2013, 5, 387–396.
- Sankaranarayanan, R.; Nene, B.M.; Shastri, S.S.; Jayant, K.; Muwonge, R.; Budukh, A.M.; Hingmire, S.; Malvi, S.G.; Thorat, R.; Kothari, A.; et al. HPV screening for cervical cancer in rural India. N. Engl. J. Med. 2009, 360, 1385–1394.
- Steinhubl, S.R.; Muse, E.D.; Topol, E.J. The emerging field of mobile health. Sci. Transl. Med. 2015, 7, 283rv3.
- Abaza, H.; Marschollek, M. mHealth application areas and technology combinations. A comparison of literature from high and low/middle income countries. Methods Inf. Med. 2017, 56, e105–e122.
- Vashist, S.K.; Luppa, P.B.; Yeo, L.Y.; Ozcan, A.; Luong, J.H.T. Emerging technologies for next-generation point-of-care testing. Trends Biotechnol. 2015, 33, 692–705.
- Duclos, V.; Ye, M.; Kagoné, M.; Sanou, H.; Sawadogo, N.H.; Bibeau, G.; Sié, A. Situating mobile health: A qualitative study of mHealth expectations in the rural health district of Nouna, Burkina Faso. Health Res. Policy Syst. 2017, 15, 47.
- Perry, L.; Malkin, R. Effectiveness of medical equipment donations to improve health systems: How much medical equipment is broken in the developing world? Med. Biol. Eng. Comput. 2011, 49, 719–722.
- Pham, M.D.; Agius, P.A.; Romero, L.; McGlynn, P.; Anderson, D.; Crowe, S.M.; Luchters, S. Acceptability and feasibility of point-of-care CD4 testing on HIV continuum of care in low and middle income countries: A systematic review. BMC Health Serv. Res. 2016, 16, 343.
- Global Health Workforce Alliance; WHO. The Cost-Effectiveness of Close-to-Community Health Programmes: What do We Know and Where Are the Gaps?
- WHO. Treat. Train. Retain-Task Shifting: Global Recommendations and Guidelines; WHO Document Production Services: Geneva, Switzerland, 2008.
- Scheffler, R.M.; Liu, J.X.; Kinfu, Y.; Poz, M.R.D. Forecasting the global shortage of physicians: An economic-and needs-based approach. Bull. World Health Organ. 2008, 86, 516–523.
- Ritchie, L.M.P.; Van Lettow, M.; Makwakwa, A.; Chan, A.K.; Hamid, J.S.; Kawonga, H.; Martiniuk, A.; Schull, M.J.; Van Schoor, V.; Zwarenstein, M.; et al. The impact of a knowledge translation intervention employing educational outreach and a point-of-care reminder tool vs standard lay health worker training on tuberculosis treatment completion rates: Study protocol for a cluster randomized controlled trial. Trials 2016, 17, 439.
- Ritchie, L.M.P.; Van Lettow, M.; Barnsley, J.; Chan, A.K.; Joshua, M.; Martiniuk, A.; Schull, M.J.; Zwarenstein, M. Evaluation of lay health workers' needs to effectively support anti-tuberculosis treatment adherence in Malawi. Int. J. Tuberc. Lung Dis. 2012, 16, 1492–1497.
- Schroeder, L.F.; Elbireer, A.; Jackson, J.B.; Amukele, T.K. Laboratory diagnostics market in East Africa: A survey of test types, test availability, and test prices in Kampala, Uganda. PLoS ONE 2015, 10, e0134578.
- Mabey, D.; Sollis, K.A.; Kelly, H.A.; Benzaken, A.S.; Bitarakwate, E.; Changalucha, J.; Chen, X.-S.; Yin, Y.-P.; Garcia, P.; Strasser, S.; et al. Point-of-care tests to strengthen health systems and save newborn lives: The case of Syphilis. PLoS Med. 2012, 9, e1001233.
- Klatman, E.L.; Jenkins, A.J.; Ahmadi, M.Y.; Ogle, G.D. Blood glucose meters and test strips: Global market and challenges to access in low-resource settings. Lancet Diabetes Endocrinol. 2019, 7, 150–160.
